Welcoming NovaSonix Healthcare to the Medical Innovation Xchange Community!
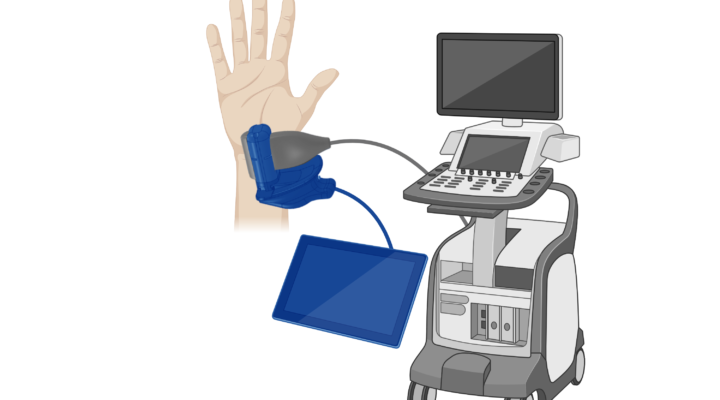
NovaSonix Healthcare has joined MIX, becoming the newest member of Canada’s growing community of med‑tech innovators. Based in London, Ontario, NovaSonix enters MIX at a pivotal stage as it advances MSKNovaVue™, a breakthrough technology that transforms any standard ultrasound probe into a real‑time 3D imaging device, delivering MRI‑level insight without the cost or wait times of traditional MRI.